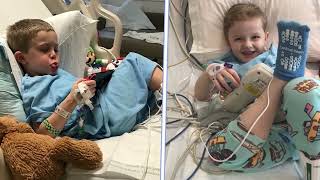
FDA Approves First Gene Therapy Treatment for Duchenne Muscular Dystrophy Developed at the Abigail Wexner Research Institute at Nationwide Children’s Hospital
Nationwide Children's HospitalToday, the Abigail Wexner Research Institute at Nationwide Children’s Hospital praised the Food and Drug Administration (FDA) for its accelerated approval of SRP-9001/ELEVIDYS for Duchenne muscular dystrophy (DMD) following decades of research in its Center for Gene Therapy to help patients with neuromuscular diseases.